Consider the potential of one-time gene replacement therapy for your patients and practice
Dosing characteristics
ITVISMA is the only single-dose treatment approved for patients aged 2 years and older with spinal muscular atrophy (SMA)1
Dosing and administration of SMA treatments for patients aged 2 years and older1-3 | |||
Treatment | ITVISMA | SPINRAZA® (nusinersen) | EVRYSDI® |
Dosing frequency | A single dosea | Maintenance doses: every 4 months (after 4 loading doses across 72-day treatment onboarding/initiation period) | Once daily |
Route of administration | Intrathecal injection | Intrathecal injection | Orally as liquid or tablet |
Premedication | Yes | No | No |
Immediate | Yes | Yes | No |
This chart is intended to show dosing and administration information only. Please refer to each product’s specific prescribing information for complete dosing and administration instructions. No conclusions of comparative efficacy or safety should be drawn.
aMonitor liver function (ALT, AST, total bilirubin) for at least 3 months following ITVISMA injection, and at other times as clinically indicated. Promptly assess and closely monitor patients with worsening liver function test results and/or signs or symptoms of acute illness.1
EVRYSDI is a registered trademark of Genentech USA, Inc. SPINRAZA is a registered trademark of Biogen.
Mechanism of action
ITVISMA is designed to work continuously, addressing the root cause of SMA by introducing a functional copy of SMN11
SMA is a rare genetic disease caused by deletions or mutations of the survival motor neuron 1 gene, or SMN1, which is critical for normal function of motor neurons.4-6
ITVISMA is an adeno-associated, viral vector-based gene replacement therapy designed to continuously produce survival motor neuron (SMN) protein through a one-time, single lumbar injection.1
Patients will need to receive an oral corticosteroid before and after administration of ITVISMA. Patients will also need to undergo blood tests before and for at least 3 months after administration.1
See the Before Treatment page for more details.
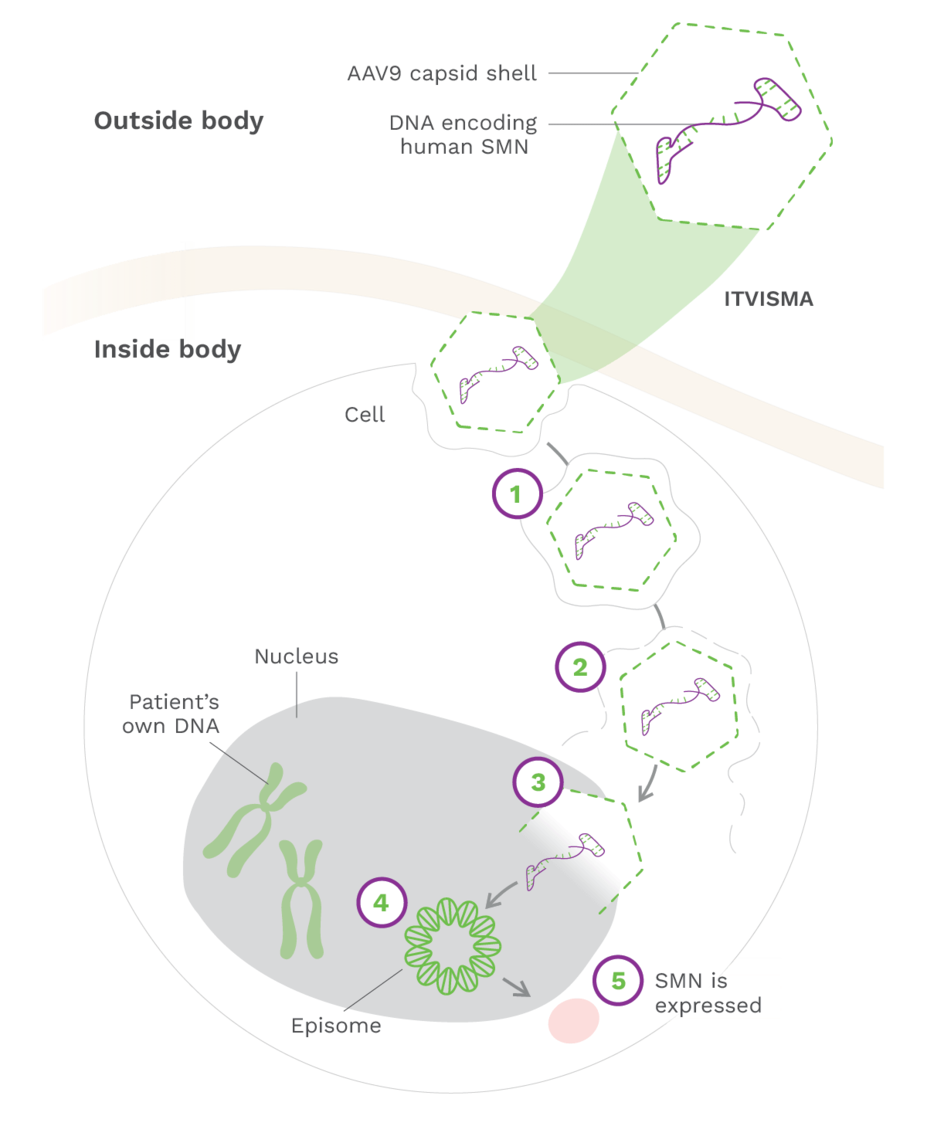
How ITVISMA works1,7
1. The adeno-associated virus serotype 9 (AAV9) vector enters motor neurons1
2. The AAV9 vector delivers the survival motor neuron gene (SMN) to the cell nucleus1,7
3. SMN is introduced to target cells as recombinant, self-complementary DNA1
4. The self-complementary ends form a circular episome that can persist in the nucleus of motor neuron cells. These cells are nondividing7,8
5. This results in rapid activation and continuous expression of SMN, leading to the production of SMN protein9