STRENGTH: supportive safety study in previously treated patients with SMA1
STRENGTH study design
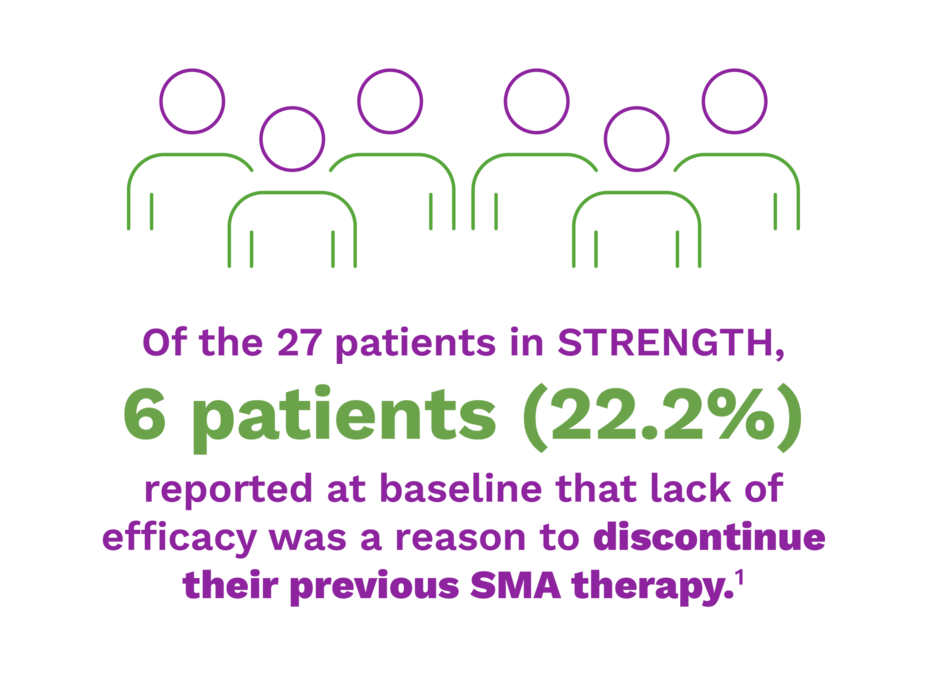
STRENGTH was an open-label, single-arm, multicenter, phase 3b, safety study with efficacy as a secondary endpoint in 27 patients with spinal muscular atrophy (SMA), aged 2 to <18 years, who discontinued previous SMA treatment and were able to sit independently* but have never taken steps independently.1
*Independent sitting is defined as sitting up straight with the head erect for at least 10 seconds without using arms or hands to balance the body or support the position.1
HFMSE score
HFMSE scores over 52 weeks with ITVISMA1
Change from baseline to Week 52 in HFMSE total score for the overall study1
Data are displayed as mean (SD) and LS mean (95% CI).1
As a secondary endpoint, the mean (SD) change from baseline in Hammersmith Functional Motor Scale — Expanded (HFMSE) score at Week 52 was 0.17 (2.88) for patients treated with ITVISMA (n=21)1
Additionally, as an exploratory endpoint, the LS mean change from baseline in HFMSE score was 1.05 (95% CI: –0.21, 2.32) at Week 52, using the MMRM method (n=21)1
RULM score
RULM score over 52 weeks with ITVISMA1
Change from baseline to Week 52 in RULM total score for the overall study1
Data are displayed as mean (SD) and LS mean (95% CI).1
As a secondary endpoint, the mean (SD) change from baseline in RULM score at Week 52 was 0.29 (2.85) for ITVISMA (n=21)1
Additionally, as an exploratory endpoint, the LS mean change from baseline in RULM score was 0.59 (95 % CI: -0.56, 1.73) at Week 52, using the MMRM method (n=21)1